
Case Study: Inside a Journey of IHC Assay Development
|
Discover
FDA Veteran Paz Vellanki, MD, joins Precision to become one of 3 ex-FDA oncology leaders supporting clients
false
100% sourced from leading minds on the front lines of research


has not third author: true, (SizeLimitingPyMap: {main={hs_id=209983237513, hs_child_table_id=0, hs_updated_at=1774533604788, hs_published_at=1779284761743, description=Calvin Niu received his BD in pharmaceuticals from Lanzhou University. He has brought over 22 years of drug development and regulatory experience in pharmaceutical companies and CROs. Calvin’s regulatory expertise covers regulatory agency engagements and all types of regulatory submissions throughout product development and post market life cycles in oncology, immunology, ophthalmology, clinical nutrition, cardiovascular, gastroenterology, women’s health, CNS, rare disease, etc. , avatar=Image{width=276,height=290,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Calvin%20Niu.png',altText='Calvin Niu',fileId=209991702802}, lastname=Niu, hs_initial_published_at=1774533614867, hs_created_by_user_id=51739740, hs_created_at=1774531720533, hs_is_edited=false, hs_deleted_at=0, name=Calvin, job=Director, Regulatory Affairs, China, slug=calvin-niu, hs_updated_by_user_id=51739740}, second={}, third={}})

Calvin Niu

has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778858, hs_child_table_id=0, hs_updated_at=1754641583843, hs_published_at=1779284761743, description=Rob Maiale is a marketing strategist and creative technologist with 17 years of experience turning complex ideas into growth. He currently leads content strategy, where he transforms insights from Precision experts into market-shaping narratives that drive the advancement of next-generation therapies. Rob’s career spans journalism, advertising, and brand storytelling, with a through-line of making the meaningful memorable. His work blends creative strategy with emerging AI tools to help Precision stay ahead of the curve—and above the noise., avatar=Image{width=287,height=287,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/PfMxMarkentive/authors/rob.png',altText='rob',fileId=194099000843}, linkedin=https://www.linkedin.com/in/rob-maiale/, hs_name=, hs_path=, lastname=Maiale, hs_initial_published_at=1775839468505, hs_created_at=1709645745089, hs_is_edited=false, hs_deleted_at=0, name=Rob, position=Associate Director, Digital Content & Inbound Marketing, job=, slug=rob-maiale, email=, hs_updated_by_user_id=51739740}, second={}, third={}})

Rob Maiale

has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778843, hs_child_table_id=0, hs_updated_at=1774960706353, hs_published_at=1779284761743, description=Robert Bauer is an Executive Director of Operational Strategy at Precision with over 20 years of clinical trial operations experience from both the sponsor and CRO perspectives. He has led teams in the conduct of trials from Phase I through IV and has experience across a wide variety of trial designs. Bob has experience across many therapeutic areas some of which include rare diseases, oncology, depression, schizophrenia, and substance use disorders., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Robert%20Bauer%20Square.webp',altText='Robert Bauer Square',fileId=165892429961}, linkedin=https://www.linkedin.com/in/robert-bauer-a78b704b, hs_name=, hs_path=, lastname=Bauer, hs_initial_published_at=1775839468505, hs_created_at=1709645745074, hs_is_edited=false, hs_deleted_at=0, name=Robert, position=Vice President, Operational Strategy, job=, slug=robert-bauer, email=, hs_updated_by_user_id=51739740}, second={hs_id=185416008260, hs_child_table_id=0, hs_updated_at=1754641208207, hs_published_at=1779284761743, description=Sarika is a Senior Clinical Trial Manager with 14+ years of experience in clinical research, specializing in oncology, rare diseases, and cardiology. With a robust focus on Phase I-IV clinical trials, she has successfully led projects from start-up to close-out and has experience in cohort management and solid tumor studies. She is a strong advocate for cross-functional teamwork, streamlining processes, and optimizing project efficiency., avatar=Image{width=287,height=287,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/PfMxMarkentive/authors/Sarika-David-Armogan-1-1.png',altText='Sarika-David-Armogan-1-1',fileId=194123711873}, lastname=David-Armogan, hs_initial_published_at=1737992355064, hs_created_by_user_id=26433386, hs_created_at=1737992180108, hs_is_edited=false, hs_deleted_at=0, name=Sarika, slug=sarika-david-armogan, hs_updated_by_user_id=51739740}, third={}})

Robert B.

Sarika D.

has not third author: true, (SizeLimitingPyMap: {main={hs_id=199448088864, hs_child_table_id=0, hs_updated_at=1762810484868, hs_published_at=1779284761743, avatar=Image{width=1200,height=1200,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Kaylee%20Mueller.png',altText='Kaylee Mueller',fileId=199456336149}, lastname=Mueller, hs_initial_published_at=1762810487482, hs_created_by_user_id=26433386, hs_created_at=1762810467146, hs_is_edited=false, hs_deleted_at=0, name=Kaylee, slug=kaylee-mueller, hs_updated_by_user_id=51739740}, second={}, third={}})

Kaylee Mueller

Translational Research - Biospecimens - Oncology
has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778886, hs_child_table_id=0, hs_updated_at=1763657611624, hs_published_at=1779284761743, description=Robert Snyder, PhD, is a seasoned expert with a 20-year track record in genomics, specializing in in vitro diagnostic assay development and bioinformatics. Committed to advancing medical science, he is proficient in regulatory V&V study designs and understands the nuances of orthogonal methods. A collaborative partner in biomedical development, Dr. Snyder champions the critical transition of biospecimens from benchtop to bedside., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Robert%20Snyder.webp',altText='Robert Snyder',fileId=165892444501}, linkedin=https://www.linkedin.com/in/robert-snyder-ph-d-9b67b035, hs_name=, hs_path=, lastname=Snyder, PhD, hs_initial_published_at=1775839468505, hs_created_at=1709645745117, hs_is_edited=false, hs_deleted_at=0, name=Robert, job=, slug=robert-snyder, email=, hs_updated_by_user_id=51739740}, second={hs_id=164691024166, hs_child_table_id=0, hs_updated_at=1716326265678, hs_published_at=1779284761743, description=Respected veteran of the biospecimen and biomarker laboratory services industry. Honed his expertise through leadership roles in organizations including Roche, Cancer Genetics, and BioServe. Holds a Master of Public Health and a Masters of Business Administration from the Johns Hopkins University., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Rob%20Fannon%20Headshot.webp',altText='Rob Fannon Headshot',fileId=165893006477}, linkedin=https://www.linkedin.com/in/rob-fannon-901378/, team=[{id=163072475009, name='Translational Sciences Leadership'}], lastname=Fannon, MPH, MBA, hs_initial_published_at=1713346931329, hs_created_by_user_id=62155952, hs_created_at=1713345787919, hs_is_edited=false, hs_deleted_at=0, name=Rob, job=General Manager, Biospecimen Solutions, slug=rob-fannon, hs_updated_by_user_id=51739740}, third={}})

Robert S.

Rob F.

Translational Research - Biospecimens
has not third author: true, (SizeLimitingPyMap: {main={hs_id=199448088864, hs_child_table_id=0, hs_updated_at=1762810484868, hs_published_at=1779284761743, avatar=Image{width=1200,height=1200,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Kaylee%20Mueller.png',altText='Kaylee Mueller',fileId=199456336149}, lastname=Mueller, hs_initial_published_at=1762810487482, hs_created_by_user_id=26433386, hs_created_at=1762810467146, hs_is_edited=false, hs_deleted_at=0, name=Kaylee, slug=kaylee-mueller, hs_updated_by_user_id=51739740}, second={}, third={}})

Kaylee Mueller

has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778874, hs_child_table_id=0, hs_updated_at=1765824406763, hs_published_at=1779284761743, description=Kennon Daniels is a scientific and regulatory in vitro diagnostic authority, specializing in the development of US regulatory strategies and FDA marketing submissions for IVD assays and instruments. Her expertise spans all aspects of product development, analytical and clinical trial protocol designs, Q-Submissions, IDEs, de novos, 510(k)s, and pre-market approval platforms., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Kennon%20Daniels.webp',altText='Kennon Daniels',fileId=165893120790}, linkedin=https://www.linkedin.com/in/kennon-daniels-ph-d-84a90b6, hs_name=, hs_path=, lastname=Daniels, PhD, hs_initial_published_at=1775839468505, hs_created_at=1709645745105, hs_is_edited=false, hs_deleted_at=0, name=Kennon, position=Vice President, IVD Regulatory Consulting, job=, slug=kennon-daniels, email=, hs_updated_by_user_id=51739740}, second={hs_id=164426546714, hs_child_table_id=0, hs_updated_at=1778171432595, hs_published_at=1779284761743, description=Angélique Heckmann, PhD is a people‑driven leader who combines scientific expertise and business acumen with a strong foundation in human values. With a background in therapeutic drug development and an MBA‑shaped strategic mindset, she focuses on building meaningful, long‑term relationships with teams, partners, and clients. By bridging science and strategy, she supports informed decision‑making, fosters collaboration, and helps translate innovation into real‑world impact—always guided by a shared purpose: advancing solutions that ultimately improve patients’ lives, avatar=Image{width=287,height=287,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/PfMxMarkentive/authors/angelique-1.png',altText='angelique-1',fileId=194126030110}, lastname=Heckmann, PhD, hs_initial_published_at=1713174153602, hs_created_by_user_id=62155952, hs_created_at=1713172107894, hs_is_edited=false, hs_deleted_at=0, name=Angelique, job=Regional Vice President, Translational Business Solutions, slug=angelique-heckmann, hs_updated_by_user_id=51739740}, third={}})

Kennon D.

Angelique H.
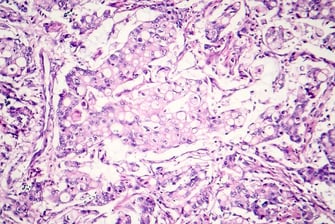
Translational Research - Regulatory - Biomarkers - Assays
has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778874, hs_child_table_id=0, hs_updated_at=1765824406763, hs_published_at=1779284761743, description=Kennon Daniels is a scientific and regulatory in vitro diagnostic authority, specializing in the development of US regulatory strategies and FDA marketing submissions for IVD assays and instruments. Her expertise spans all aspects of product development, analytical and clinical trial protocol designs, Q-Submissions, IDEs, de novos, 510(k)s, and pre-market approval platforms., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Kennon%20Daniels.webp',altText='Kennon Daniels',fileId=165893120790}, linkedin=https://www.linkedin.com/in/kennon-daniels-ph-d-84a90b6, hs_name=, hs_path=, lastname=Daniels, PhD, hs_initial_published_at=1775839468505, hs_created_at=1709645745105, hs_is_edited=false, hs_deleted_at=0, name=Kennon, position=Vice President, IVD Regulatory Consulting, job=, slug=kennon-daniels, email=, hs_updated_by_user_id=51739740}, second={hs_id=182710436924, hs_child_table_id=0, hs_updated_at=1754641257915, hs_published_at=1779284761743, description=Christie Bongel, MS, CT(ASCP), brings nearly two decades of experience in both research and clinical medicine. As the site director for Precision for Medicine’s IHC Center of Excellence in North Carolina, she excels in managing scientific operations and collaborating with clients. Christie is skilled in cytology, laboratory medicine, and molecular diagnostics. Her role focuses on building an energized team dedicated to delivering life-changing outcomes for patients. Christie earned her MS in Applied Biotechnology and is an ASCP certified cytotechnologist., avatar=Image{width=287,height=287,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/PfMxMarkentive/authors/Christie-Bongel_Square_260px-1.png',altText='Christie-Bongel_Square_260px-1',fileId=194123799456}, lastname= Bongel, hs_initial_published_at=1731517828480, hs_created_by_user_id=28386183, hs_created_at=1731419696820, hs_is_edited=false, hs_deleted_at=0, name=Christie, job=Site Director - Histology, slug=christie-bongel, hs_updated_by_user_id=51739740}, third={}})

Kennon D.

Christie .

Translational Research - Regulatory
has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778874, hs_child_table_id=0, hs_updated_at=1765824406763, hs_published_at=1779284761743, description=Kennon Daniels is a scientific and regulatory in vitro diagnostic authority, specializing in the development of US regulatory strategies and FDA marketing submissions for IVD assays and instruments. Her expertise spans all aspects of product development, analytical and clinical trial protocol designs, Q-Submissions, IDEs, de novos, 510(k)s, and pre-market approval platforms., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Kennon%20Daniels.webp',altText='Kennon Daniels',fileId=165893120790}, linkedin=https://www.linkedin.com/in/kennon-daniels-ph-d-84a90b6, hs_name=, hs_path=, lastname=Daniels, PhD, hs_initial_published_at=1775839468505, hs_created_at=1709645745105, hs_is_edited=false, hs_deleted_at=0, name=Kennon, position=Vice President, IVD Regulatory Consulting, job=, slug=kennon-daniels, email=, hs_updated_by_user_id=51739740}, second={hs_id=159488778882, hs_child_table_id=0, hs_updated_at=1714461102447, hs_published_at=1779284761743, description=Dr. Curnutte has more than ten years of research and regulatory affairs experience focused on in vitro diagnostics (IVDs). She has held regulatory positions at both device and pharmaceutical companies, during which she led seven U.S. and ex-U.S. companion diagnostic registrations. She has extensive expertise in CDx development and clinical trial integration, IVD product development, next-generation sequencing technologies, and translation of U.S. regulatory strategy to major markets., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Margaret%20Curnutte.webp',altText='Margaret Curnutte',fileId=165892782094}, linkedin=https://www.linkedin.com/in/margaret-curnutte-93558913, hs_name=, hs_path=, lastname=Curnutte, hs_initial_published_at=1775839468505, hs_created_at=1709645745113, hs_is_edited=false, hs_deleted_at=0, name=Margaret, job=, slug=margaret-curnutte, email=, hs_updated_by_user_id=51739740}, third={}})

Kennon D.

Margaret C.

Translational Research - Regulatory - Gene Therapies
has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778874, hs_child_table_id=0, hs_updated_at=1765824406763, hs_published_at=1779284761743, description=Kennon Daniels is a scientific and regulatory in vitro diagnostic authority, specializing in the development of US regulatory strategies and FDA marketing submissions for IVD assays and instruments. Her expertise spans all aspects of product development, analytical and clinical trial protocol designs, Q-Submissions, IDEs, de novos, 510(k)s, and pre-market approval platforms., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Kennon%20Daniels.webp',altText='Kennon Daniels',fileId=165893120790}, linkedin=https://www.linkedin.com/in/kennon-daniels-ph-d-84a90b6, hs_name=, hs_path=, lastname=Daniels, PhD, hs_initial_published_at=1775839468505, hs_created_at=1709645745105, hs_is_edited=false, hs_deleted_at=0, name=Kennon, position=Vice President, IVD Regulatory Consulting, job=, slug=kennon-daniels, email=, hs_updated_by_user_id=51739740}, second={hs_id=159488778882, hs_child_table_id=0, hs_updated_at=1714461102447, hs_published_at=1779284761743, description=Dr. Curnutte has more than ten years of research and regulatory affairs experience focused on in vitro diagnostics (IVDs). She has held regulatory positions at both device and pharmaceutical companies, during which she led seven U.S. and ex-U.S. companion diagnostic registrations. She has extensive expertise in CDx development and clinical trial integration, IVD product development, next-generation sequencing technologies, and translation of U.S. regulatory strategy to major markets., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Margaret%20Curnutte.webp',altText='Margaret Curnutte',fileId=165892782094}, linkedin=https://www.linkedin.com/in/margaret-curnutte-93558913, hs_name=, hs_path=, lastname=Curnutte, hs_initial_published_at=1775839468505, hs_created_at=1709645745113, hs_is_edited=false, hs_deleted_at=0, name=Margaret, job=, slug=margaret-curnutte, email=, hs_updated_by_user_id=51739740}, third={}})

Kennon D.

Margaret C.

has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778874, hs_child_table_id=0, hs_updated_at=1765824406763, hs_published_at=1779284761743, description=Kennon Daniels is a scientific and regulatory in vitro diagnostic authority, specializing in the development of US regulatory strategies and FDA marketing submissions for IVD assays and instruments. Her expertise spans all aspects of product development, analytical and clinical trial protocol designs, Q-Submissions, IDEs, de novos, 510(k)s, and pre-market approval platforms., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Kennon%20Daniels.webp',altText='Kennon Daniels',fileId=165893120790}, linkedin=https://www.linkedin.com/in/kennon-daniels-ph-d-84a90b6, hs_name=, hs_path=, lastname=Daniels, PhD, hs_initial_published_at=1775839468505, hs_created_at=1709645745105, hs_is_edited=false, hs_deleted_at=0, name=Kennon, position=Vice President, IVD Regulatory Consulting, job=, slug=kennon-daniels, email=, hs_updated_by_user_id=51739740}, second={}, third={}})

Kennon Daniels, PhD

has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778874, hs_child_table_id=0, hs_updated_at=1765824406763, hs_published_at=1779284761743, description=Kennon Daniels is a scientific and regulatory in vitro diagnostic authority, specializing in the development of US regulatory strategies and FDA marketing submissions for IVD assays and instruments. Her expertise spans all aspects of product development, analytical and clinical trial protocol designs, Q-Submissions, IDEs, de novos, 510(k)s, and pre-market approval platforms., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Kennon%20Daniels.webp',altText='Kennon Daniels',fileId=165893120790}, linkedin=https://www.linkedin.com/in/kennon-daniels-ph-d-84a90b6, hs_name=, hs_path=, lastname=Daniels, PhD, hs_initial_published_at=1775839468505, hs_created_at=1709645745105, hs_is_edited=false, hs_deleted_at=0, name=Kennon, position=Vice President, IVD Regulatory Consulting, job=, slug=kennon-daniels, email=, hs_updated_by_user_id=51739740}, second={}, third={}})

Kennon Daniels, PhD