Patient Experiences When Searching for Information on MEN1
Multiple endocrine neoplasia 1 (MEN1) is a rare inherited pathology that occurs in one of 30,000 people. Scientists have identified hundreds of different mutations of the MEN1 gene that can cause the disorder. It is caused by inactivating germline mutations in the MEN1 gene on chromosome 11 (11q13), encoding the menin protein which is a tumor suppressor protein.
The etiology is unknown in most cases. The most common tumors seen in patients with MEN1 involve the parathyroid gland, islet cells of the pancreas, and pituitary gland. Other endocrine tumors seen in this population include adrenal cortical tumors, neuroendocrine tumors, and pheochromocytomas. Although most tumors in people with MEN1 are benign, approximately 1 of 3 pancreatic neuroendocrine tumors and mediastinal neuroendocrine tumors are cancerous.1
The diagnosis of MEN1 is performed in two different contexts:
- People with no previous family history of MEN1 who present with at least two of the most common tumors (e.g., parathyroid tumor, pancreatic neuroendocrine tumor, or pituitary gland tumor)
- People with a previous family history of MEN1 who are diagnosed with one of the most common tumors (e.g., parathyroid tumor, pancreatic neuroendocrine tumor, or pituitary gland tumor)
This case study is focused on a targeted search for information on MEN1, assuming the search is conducted by a Spanish born, English proficient healthcare professional living in Spain with a family member recently diagnosed with MEN1. The targeted search was conducted both in English and in Spanish to assess the weight of language on access to information.
English Language Search Results for MEN1
A simple search in English for “Multiple Endocrine Neoplasia Type 1” on Google delivers 17.6 million results. It was time-consuming and cumbersome to distinguish reliable or useful information from this search. Results were divided across three different categories:
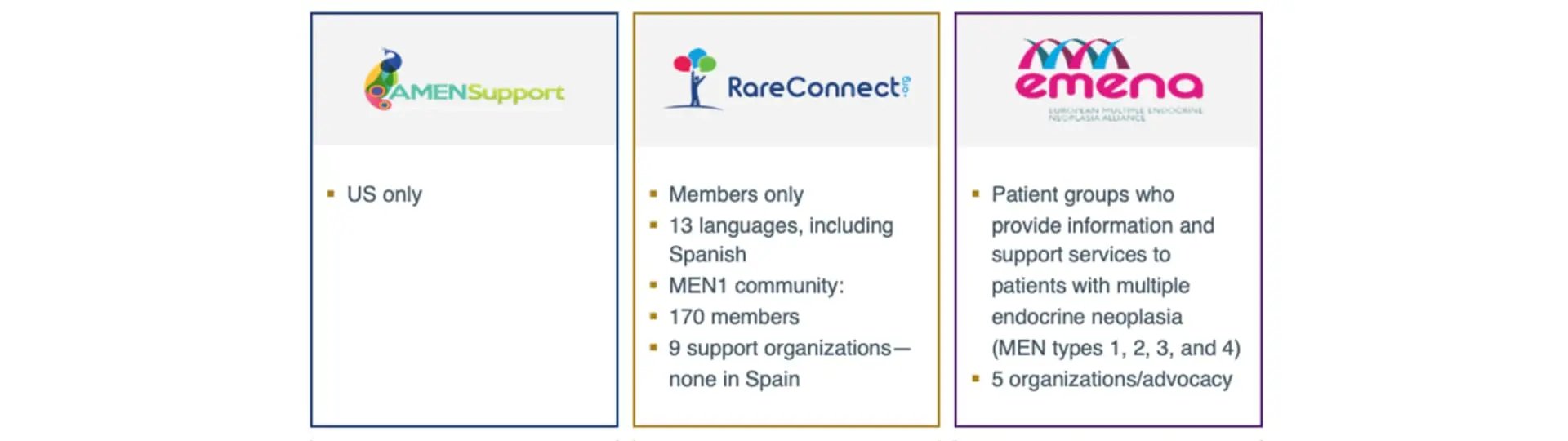
- MEN1 advocacy groups based in the U.S. only, with information exclusively released in English, like American Multiple Endocrine Neoplasia Support
- Organizations with a broader scope, like RareConnect, which releases reliable information in 13 different languages, including Spanish, but requires a membership to access to more tailored opportunities (e.g., A patient community with 170+ members and 9 support organizations)
- European-centric organizations, like European Men Alliance (EMENA), which embodied by individual patient advocacy groups who provide information about MEN disorders—none of which are in Spain
Some of these organizations link to clinicaltrials.gov, indicating the next logical step would be to look for information about MEN1 clinical studies. This search rendered 15 results. When looking more in depth, only three of these studies were truly interventional ones, and none of them had investigational sites in Spain.
Spanish Language Search Results for MEN1
The same simple Google search was performed in Spanish. Here, the search rendered more than 215,000 results—a number still unfeasible for an accurate and useful interpretation. Two websites were identified that released high-quality information in Spanish—Orphanet and FEDER (Spanish Federation of Rare Diseases)—but these were about rare diseases in general. Neither site referred to MEN1. The final approach was to conduct a Facebook search. While this did render three private patient groups, none were Spanish language facing, nor did they include Spanish members.

There is a clear information barrier between the languages
Our journey searching for MEN1 trials was not unlike what many others may face on the quest for information. Search results varied greatly between the two languages, as did the type of information served by the search engine. Both the English and Spanish search results pulled a variety of advocacy groups (some of which truly focused on the MEN1 community), but the only language in which users could interact was English. This shows the limited availability of information for non-English speaking individuals.
Driving equality in rare disease clinical research begins with improving access to information
Rare disease clinical trials will continue to be more complex than other therapeutic areas, costing patients more time for research and sponsors additional time and resources for recruitment and site set-up. As long as difficulties in locating information persist, patients will need to be proactive in managing care.
One of the most important lessons the COVID-19 pandemic taught us about clinical trial accessibility, is that research efforts must be aligned to patient needs. Leveraging decentralized clinical trial strategies and showing flexibility in study protocol implementation can reduce patient burden and improve retention—but our responsibilities run deeper.
Healthcare professionals and patient advocacy groups alike must realize that diversity and inclusiveness begin with the dissemination of information in languages other than English.
Es nuestra responsabilidad, como profesionales de la salud y grupos de apoyo a pacientes, el comprender que la diversidad y la inlcusividad comienza con diseminar la información en otros idiomas aparte del inglés
Precision Subject Matter Expert
By improving access to information, patients can be empowered to take a more proactive role in their own care, becoming partners instead of mere participants.
References:
1. Cancer.net. Multiple endocrine neoplasia type 2. American Society of Clinical Oncology (ASCO). https://www.cancer.net/cancer-types/multiple-endocrine-neoplasia-type-1. Accessed June 30, 2022.





