
Clinical Logistics Services
Precision medicine and biomarker-driven clinical trials pose a set of challenges unique from classical clinical trials, and require unique clinical logistics services solutions.
The complexity inherent in kit development, logistics, sample management, and data requires an approach to clinical logistics services that is systematic, robust, and can simplify clinical trial management: a translational clinical logistics services solution. Precision for Medicine offers clinical logistics services in support of global trials including kitting, logistics, sample processing, biostorage, and related biospecimen data services. In addition, Precision offers a comprehensive suite of specialty lab services including flow cytometry, genomics and NGS, bioanalysis, and tissue and liquid biopsy analysis.
Clinical Logistics Services designed for biomarker-driven clinical trials
Precision’s harmonized approach is rooted in a deep understanding of the needs of complex biomarker trials—where excellence in customized kit development, logistics, data management, and technology are necessary.
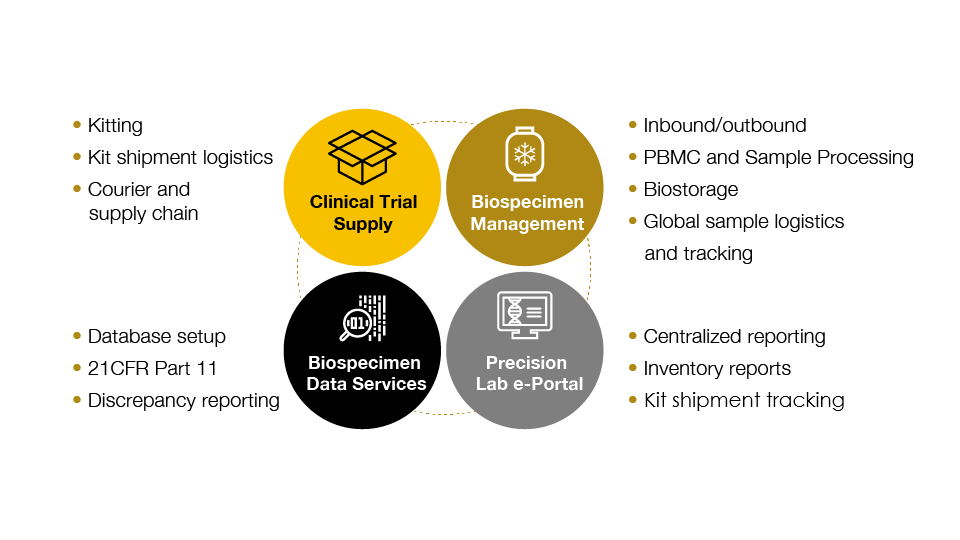
Advanced program management drives rapid project startup and study success
A clear program management structure is critical in order to consistently execute on all aspects of a clinical logistics services project. Precision’s approach integrates all aspects of a central lab service project with a single point of contact and provides:
- Dedicated study startup structure
- Document creation
- QA-approved lab manual creation
- Site training documentation
- Designated project manager as the point of contact
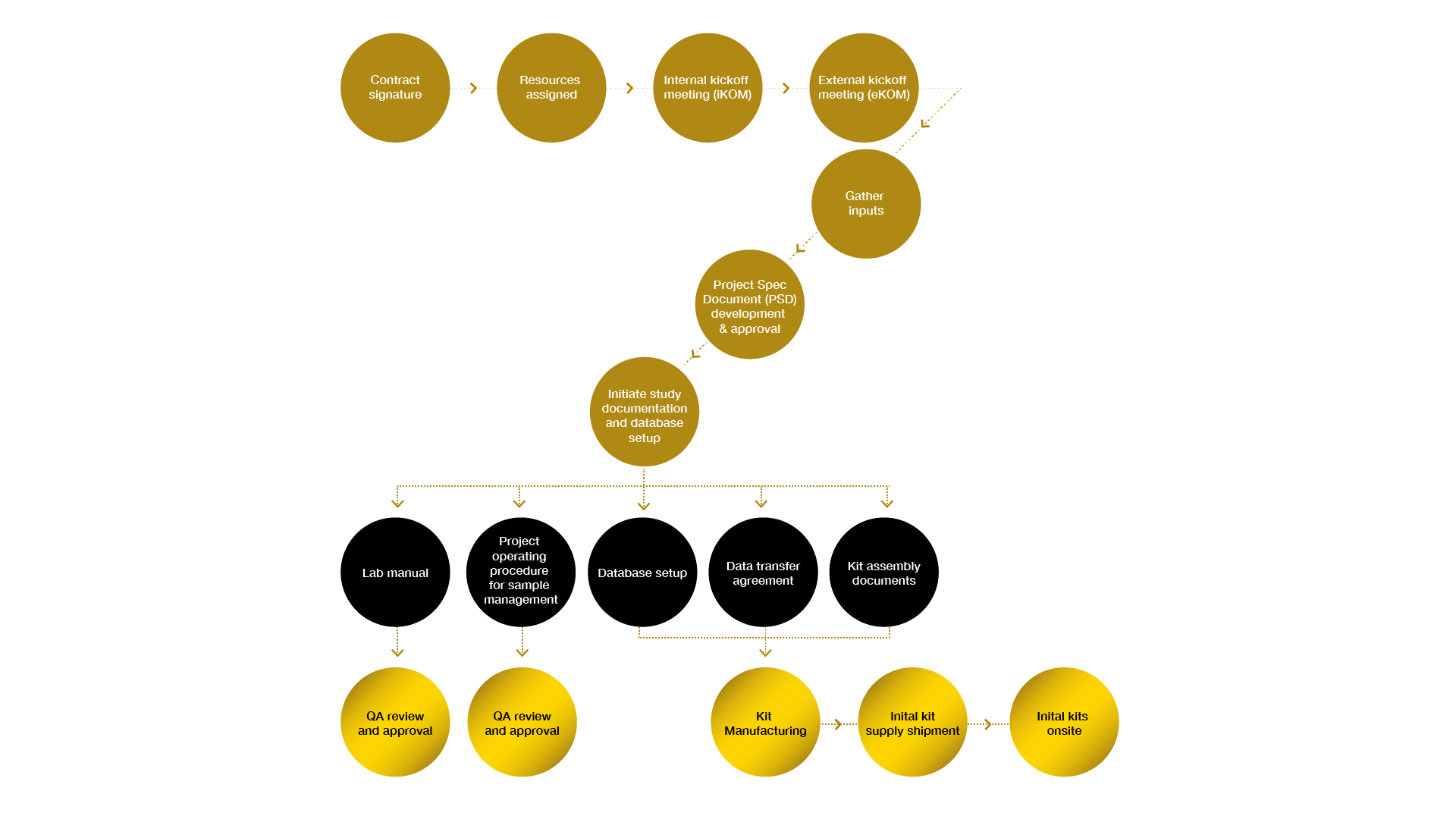
Well-structured and coordinated processes for project initiation promote a successful startup with rapid study startup timelines.
Project initiation workflow
Expansive global reach ensures timely and consistent sample processing and analysis
Clinical Reach
Translational Clinical Logistics Services: Solving the complex challenges of biomarker-driven clinical trials






