
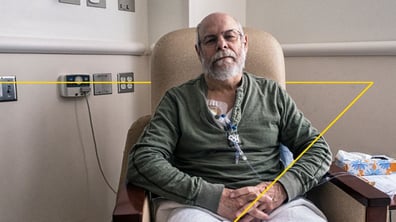
Taiwan’s Clinical Trial Landscape: Fast, Efficient, and High-Quality Research
|
Discover
false
100% sourced from leading minds on the front lines of research


has third author: false, (SizeLimitingPyMap: {main={hs_id=167906611101, hs_child_table_id=0, hs_updated_at=1743507158804, hs_published_at=1752765086448, description=Helen brings over 30 years of experience in multinational healthcare, biotech, and pharmaceutical industries, including work with Medical Research Institutes and CROs. She specializes in leading clinical research, drug and device development, and fostering innovative research collaborations. Based in Sydney, Australia, Helen has a strong track record of delivering clinical trials across the APAC region., avatar=Image{width=600,height=700,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Helene-Ormandy.png',altText='Helene-Ormandy',fileId=168387434767}, lastname=Ormandy, PhD, hs_initial_published_at=1716212950394, hs_created_by_user_id=2737751, hs_created_at=1716212897654, hs_is_edited=false, hs_deleted_at=0, name=Helen, job=Senior Director, Clinical Operations, slug=helen-ormandy, hs_updated_by_user_id=26433386}, second={hs_id=192644417560, hs_child_table_id=0, hs_updated_at=1752501506910, hs_published_at=1752765086448, description=Gavin Phan is a pharmacist based in Melbourne, Australia, who leads Precision’s APAC CRA team. Gavin has over a decade of experience in diverse clinical operations and leading teams throughout AsiaPac, including Taiwan., avatar=Image{width=622,height=624,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Gavin%20Phan.jpg',altText='Gavin Phan',fileId=192648011133}, lastname=Phan, hs_initial_published_at=1752501514571, hs_created_by_user_id=26433386, hs_created_at=1752501444306, hs_is_edited=false, hs_deleted_at=0, name=Gavin, job=Manager, Global Clinical Monitoring, slug=gavin-phan, hs_updated_by_user_id=26433386}, third={hs_id=192645526119, hs_child_table_id=0, hs_updated_at=1752501710597, hs_published_at=1752765086448, description=Kelly Lin is a Senior Clinical Research Associate supporting Precision clinical trials in the AsiaPac region., avatar=Image{width=1200,height=1200,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Precision%20Expert.png',altText='Precision Expert',fileId=164175466823}, lastname=Lin, hs_initial_published_at=1752501630185, hs_created_by_user_id=26433386, hs_created_at=1752501557958, hs_is_edited=false, hs_deleted_at=0, name=Kelly, job=Senior Clinical Research Associate, slug=kelly-lin, hs_updated_by_user_id=9297597}})



Helen O.
Gavin P.
Kelly L.

Clinical Trials - Oncology - Rare Diseases
has not third author: true, (SizeLimitingPyMap: {main={hs_id=164725548848, hs_child_table_id=0, hs_updated_at=1716454310949, hs_published_at=1752765086448, description=Alexis Hobbins-White is an accomplished leader who plays a pivotal role in navigating the complex landscape of global oncology and rare disease-focused clinical trials. With a career spanning over 15 years in the clinical trials industry, Alexis brings a wealth of experience across multiple therapeutic areas, showcasing her proficiency in diverse aspects of operational strategy. She specializes in early phase adaptive studies, particularly in immuno-oncology and advanced therapeutics. She has an MSc in Neuroscience and an MA in Biotechnology Law and Ethics., avatar=Image{width=150,height=150,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/PfMxMarkentive/authors/Alexis-Hobbins-White-150x150.jpeg',altText='Alexis-Hobbins-White-150x150',fileId=164710912635}, linkedin=https://www.linkedin.com/in/alexis-hobbins-white-400a1720/, lastname=Hobbins-White, hs_initial_published_at=1713362276020, hs_created_by_user_id=5166971, hs_created_at=1713361952861, hs_is_edited=false, hs_deleted_at=0, name=Alexis, job=Senior Director, Operational Strategy , slug=alexis-hobbins-white, hs_updated_by_user_id=9297597}, second={}, third={}})

Alexis Hobbins-White
Clinical Trials - Oncology - Cell Therapies
has not third author: true, (SizeLimitingPyMap: {main={hs_id=164725548848, hs_child_table_id=0, hs_updated_at=1716454310949, hs_published_at=1752765086448, description=Alexis Hobbins-White is an accomplished leader who plays a pivotal role in navigating the complex landscape of global oncology and rare disease-focused clinical trials. With a career spanning over 15 years in the clinical trials industry, Alexis brings a wealth of experience across multiple therapeutic areas, showcasing her proficiency in diverse aspects of operational strategy. She specializes in early phase adaptive studies, particularly in immuno-oncology and advanced therapeutics. She has an MSc in Neuroscience and an MA in Biotechnology Law and Ethics., avatar=Image{width=150,height=150,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/PfMxMarkentive/authors/Alexis-Hobbins-White-150x150.jpeg',altText='Alexis-Hobbins-White-150x150',fileId=164710912635}, linkedin=https://www.linkedin.com/in/alexis-hobbins-white-400a1720/, lastname=Hobbins-White, hs_initial_published_at=1713362276020, hs_created_by_user_id=5166971, hs_created_at=1713361952861, hs_is_edited=false, hs_deleted_at=0, name=Alexis, job=Senior Director, Operational Strategy , slug=alexis-hobbins-white, hs_updated_by_user_id=9297597}, second={}, third={}})

Alexis Hobbins-White

Translational Research - Biomarkers
has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778875, hs_child_table_id=0, hs_updated_at=1716454216458, hs_published_at=1752765086448, description=Pharma industry veteran and expert at biomarker-driven clinical trial design and execution. Leader of biomarker and drug development programs for pharmaceutical and diagnostics companies, as well as the National Institutes of Health. Spearheaded the discovery of pharmacodynamic biomarkers and novel targets for inflammatory disease therapy., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Deb%20Phippard.webp',altText='Deb Phippard',fileId=165892372605}, linkedin=https://www.linkedin.com/in/deb-phippard-103b54, team=[{id=163072475007, name='Precision for Medicine Leadership'}], hs_name=, hs_path=, lastname=Phippard, PhD, hs_initial_published_at=1716212769184, hs_created_at=1709645745106, hs_is_edited=false, hs_deleted_at=0, name=Deborah, job= Chief Scientific Officer, slug=deborah-phippard, email=, hs_updated_by_user_id=9297597}, second={hs_id=182710436924, hs_child_table_id=0, hs_updated_at=1731419754428, hs_published_at=1752765086448, description=Christie Bongel, MS, CT(ASCP), brings nearly two decades of experience in both research and clinical medicine. As the site director for Precision for Medicine’s IHC Center of Excellence in North Carolina, she excels in managing scientific operations and collaborating with clients. Christie is skilled in cytology, laboratory medicine, and molecular diagnostics. Her role focuses on building an energized team dedicated to delivering life-changing outcomes for patients. Christie earned her MS in Applied Biotechnology and is an ASCP certified cytotechnologist., avatar=Image{width=260,height=260,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Christie%20Bongel_Square_260px.jpg',altText='Christie Bongel_Square_260px',fileId=178044303113}, lastname= Bongel, hs_initial_published_at=1731517828480, hs_created_by_user_id=28386183, hs_created_at=1731419696820, hs_is_edited=false, hs_deleted_at=0, name=Christie, job=Site Director - Histology, slug=christie-bongel, hs_updated_by_user_id=9297597}, third={}})

Deborah P.

Christie .

Translational Research - Regulatory - Assays - Gene Therapies
has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778875, hs_child_table_id=0, hs_updated_at=1716454216458, hs_published_at=1752765086448, description=Pharma industry veteran and expert at biomarker-driven clinical trial design and execution. Leader of biomarker and drug development programs for pharmaceutical and diagnostics companies, as well as the National Institutes of Health. Spearheaded the discovery of pharmacodynamic biomarkers and novel targets for inflammatory disease therapy., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Deb%20Phippard.webp',altText='Deb Phippard',fileId=165892372605}, linkedin=https://www.linkedin.com/in/deb-phippard-103b54, team=[{id=163072475007, name='Precision for Medicine Leadership'}], hs_name=, hs_path=, lastname=Phippard, PhD, hs_initial_published_at=1716212769184, hs_created_at=1709645745106, hs_is_edited=false, hs_deleted_at=0, name=Deborah, job= Chief Scientific Officer, slug=deborah-phippard, email=, hs_updated_by_user_id=9297597}, second={hs_id=159488778889, hs_child_table_id=0, hs_updated_at=1714461111815, hs_published_at=1752765086448, description=Travis Harrison, PhD, is Vice President of Diagnostic Development at Precision for Medicine. He brings more than 20 years of bioanalytical assay experience to the Precision team with expertise in ligand-binding and cell-based assays. Travis has experience supporting clinical and nonclinical studies for a broad range of indications, with an emphasis on diagnostic assays to evaluate immune responses to gene therapies., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Travis%20Harrison.webp',altText='Travis Harrison',fileId=165893006481}, linkedin=https://www.linkedin.com/in/travis-harrison-phd-6108852/, hs_name=, hs_path=, lastname=Harrison, hs_initial_published_at=1716212769184, hs_created_at=1709645745120, hs_is_edited=false, hs_deleted_at=0, name=Travis, job=, slug=travis-harrison, email=, hs_updated_by_user_id=9297597}, third={}})

Deborah P.

Travis H.
Translational Research - Regulatory - Assays - Gene Therapies
has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778875, hs_child_table_id=0, hs_updated_at=1716454216458, hs_published_at=1752765086448, description=Pharma industry veteran and expert at biomarker-driven clinical trial design and execution. Leader of biomarker and drug development programs for pharmaceutical and diagnostics companies, as well as the National Institutes of Health. Spearheaded the discovery of pharmacodynamic biomarkers and novel targets for inflammatory disease therapy., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Deb%20Phippard.webp',altText='Deb Phippard',fileId=165892372605}, linkedin=https://www.linkedin.com/in/deb-phippard-103b54, team=[{id=163072475007, name='Precision for Medicine Leadership'}], hs_name=, hs_path=, lastname=Phippard, PhD, hs_initial_published_at=1716212769184, hs_created_at=1709645745106, hs_is_edited=false, hs_deleted_at=0, name=Deborah, job= Chief Scientific Officer, slug=deborah-phippard, email=, hs_updated_by_user_id=9297597}, second={hs_id=159488778840, hs_child_table_id=0, hs_updated_at=1716132440837, hs_published_at=1752765086448, description=Maham Ansari, MS, RAC is a Senior Director, IVD Regulatory Affairs. Her focus is on companion diagnostic co-development programs, where she leverages her regulatory expertise to secure approvals in the US, Europe and beyond for companion diagnostics with a specific focus on gene therapy. She brings more than 16 years of global regulatory leadership experience in the medical device and in vitro diagnostics industry, spanning major multinationals to small start-ups, including full life cycle management across all stages of product development, execution of global regulatory strategies, and post-market surveillance.sh, avatar=Image{width=710,height=710,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Maham_Ansari.png',altText='Maham_Ansari',fileId=167373022876}, linkedin=https://www.linkedin.com/in/maham-ansari-ms-rac-6a735739, hs_name=, hs_path=, lastname=Ansari, hs_initial_published_at=1716212769184, hs_created_at=1709645745071, hs_is_edited=false, hs_deleted_at=0, name=Maham, job=, slug=maham-ansari, email=, hs_updated_by_user_id=9297597}, third={}})

Deborah P.

Maham A.

has third author: false, (SizeLimitingPyMap: {main={hs_id=167906611101, hs_child_table_id=0, hs_updated_at=1743507158804, hs_published_at=1752765086448, description=Helen brings over 30 years of experience in multinational healthcare, biotech, and pharmaceutical industries, including work with Medical Research Institutes and CROs. She specializes in leading clinical research, drug and device development, and fostering innovative research collaborations. Based in Sydney, Australia, Helen has a strong track record of delivering clinical trials across the APAC region., avatar=Image{width=600,height=700,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Helene-Ormandy.png',altText='Helene-Ormandy',fileId=168387434767}, lastname=Ormandy, PhD, hs_initial_published_at=1716212950394, hs_created_by_user_id=2737751, hs_created_at=1716212897654, hs_is_edited=false, hs_deleted_at=0, name=Helen, job=Senior Director, Clinical Operations, slug=helen-ormandy, hs_updated_by_user_id=26433386}, second={hs_id=192644417560, hs_child_table_id=0, hs_updated_at=1752501506910, hs_published_at=1752765086448, description=Gavin Phan is a pharmacist based in Melbourne, Australia, who leads Precision’s APAC CRA team. Gavin has over a decade of experience in diverse clinical operations and leading teams throughout AsiaPac, including Taiwan., avatar=Image{width=622,height=624,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Gavin%20Phan.jpg',altText='Gavin Phan',fileId=192648011133}, lastname=Phan, hs_initial_published_at=1752501514571, hs_created_by_user_id=26433386, hs_created_at=1752501444306, hs_is_edited=false, hs_deleted_at=0, name=Gavin, job=Manager, Global Clinical Monitoring, slug=gavin-phan, hs_updated_by_user_id=26433386}, third={hs_id=192645526119, hs_child_table_id=0, hs_updated_at=1752501710597, hs_published_at=1752765086448, description=Kelly Lin is a Senior Clinical Research Associate supporting Precision clinical trials in the AsiaPac region., avatar=Image{width=1200,height=1200,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Precision%20Expert.png',altText='Precision Expert',fileId=164175466823}, lastname=Lin, hs_initial_published_at=1752501630185, hs_created_by_user_id=26433386, hs_created_at=1752501557958, hs_is_edited=false, hs_deleted_at=0, name=Kelly, job=Senior Clinical Research Associate, slug=kelly-lin, hs_updated_by_user_id=9297597}})



Helen O.
Gavin P.
Kelly L.

Translational Research - Biomarkers
has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778875, hs_child_table_id=0, hs_updated_at=1716454216458, hs_published_at=1752765086448, description=Pharma industry veteran and expert at biomarker-driven clinical trial design and execution. Leader of biomarker and drug development programs for pharmaceutical and diagnostics companies, as well as the National Institutes of Health. Spearheaded the discovery of pharmacodynamic biomarkers and novel targets for inflammatory disease therapy., avatar=Image{width=576,height=576,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/Headshots/Headshots%20no%20background/Deb%20Phippard.webp',altText='Deb Phippard',fileId=165892372605}, linkedin=https://www.linkedin.com/in/deb-phippard-103b54, team=[{id=163072475007, name='Precision for Medicine Leadership'}], hs_name=, hs_path=, lastname=Phippard, PhD, hs_initial_published_at=1716212769184, hs_created_at=1709645745106, hs_is_edited=false, hs_deleted_at=0, name=Deborah, job= Chief Scientific Officer, slug=deborah-phippard, email=, hs_updated_by_user_id=9297597}, second={hs_id=182710436924, hs_child_table_id=0, hs_updated_at=1731419754428, hs_published_at=1752765086448, description=Christie Bongel, MS, CT(ASCP), brings nearly two decades of experience in both research and clinical medicine. As the site director for Precision for Medicine’s IHC Center of Excellence in North Carolina, she excels in managing scientific operations and collaborating with clients. Christie is skilled in cytology, laboratory medicine, and molecular diagnostics. Her role focuses on building an energized team dedicated to delivering life-changing outcomes for patients. Christie earned her MS in Applied Biotechnology and is an ASCP certified cytotechnologist., avatar=Image{width=260,height=260,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Christie%20Bongel_Square_260px.jpg',altText='Christie Bongel_Square_260px',fileId=178044303113}, lastname= Bongel, hs_initial_published_at=1731517828480, hs_created_by_user_id=28386183, hs_created_at=1731419696820, hs_is_edited=false, hs_deleted_at=0, name=Christie, job=Site Director - Histology, slug=christie-bongel, hs_updated_by_user_id=9297597}, third={}})

Deborah P.

Christie .

Clinical Trials - Oncology - Rare Diseases
has not third author: true, (SizeLimitingPyMap: {main={hs_id=164725548848, hs_child_table_id=0, hs_updated_at=1716454310949, hs_published_at=1752765086448, description=Alexis Hobbins-White is an accomplished leader who plays a pivotal role in navigating the complex landscape of global oncology and rare disease-focused clinical trials. With a career spanning over 15 years in the clinical trials industry, Alexis brings a wealth of experience across multiple therapeutic areas, showcasing her proficiency in diverse aspects of operational strategy. She specializes in early phase adaptive studies, particularly in immuno-oncology and advanced therapeutics. She has an MSc in Neuroscience and an MA in Biotechnology Law and Ethics., avatar=Image{width=150,height=150,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/PfMxMarkentive/authors/Alexis-Hobbins-White-150x150.jpeg',altText='Alexis-Hobbins-White-150x150',fileId=164710912635}, linkedin=https://www.linkedin.com/in/alexis-hobbins-white-400a1720/, lastname=Hobbins-White, hs_initial_published_at=1713362276020, hs_created_by_user_id=5166971, hs_created_at=1713361952861, hs_is_edited=false, hs_deleted_at=0, name=Alexis, job=Senior Director, Operational Strategy , slug=alexis-hobbins-white, hs_updated_by_user_id=9297597}, second={}, third={}})

Alexis Hobbins-White

Clinical Trials - Oncology - Cell Therapies
has not third author: true, (SizeLimitingPyMap: {main={hs_id=164725548848, hs_child_table_id=0, hs_updated_at=1716454310949, hs_published_at=1752765086448, description=Alexis Hobbins-White is an accomplished leader who plays a pivotal role in navigating the complex landscape of global oncology and rare disease-focused clinical trials. With a career spanning over 15 years in the clinical trials industry, Alexis brings a wealth of experience across multiple therapeutic areas, showcasing her proficiency in diverse aspects of operational strategy. She specializes in early phase adaptive studies, particularly in immuno-oncology and advanced therapeutics. She has an MSc in Neuroscience and an MA in Biotechnology Law and Ethics., avatar=Image{width=150,height=150,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/PfMxMarkentive/authors/Alexis-Hobbins-White-150x150.jpeg',altText='Alexis-Hobbins-White-150x150',fileId=164710912635}, linkedin=https://www.linkedin.com/in/alexis-hobbins-white-400a1720/, lastname=Hobbins-White, hs_initial_published_at=1713362276020, hs_created_by_user_id=5166971, hs_created_at=1713361952861, hs_is_edited=false, hs_deleted_at=0, name=Alexis, job=Senior Director, Operational Strategy , slug=alexis-hobbins-white, hs_updated_by_user_id=9297597}, second={}, third={}})

Alexis Hobbins-White

has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778891, hs_child_table_id=0, hs_updated_at=1709645745122, hs_published_at=1752765086448, description=Kurt Preugschat has over 11 years of statistical experience from Phase I to Phase IV clinical trials. He is a lead statistician, providing statistical support and oversight for numerous clinical trials associated with new drug applications (NDA), supplemental new drug applications (sNDA), and medical devices. His expertise includes the development of study designs to meet complex clinical questions and the measurement of desired outcomes. In addition, Kurt has extensive experience generating statistical analyses and creating complex SAS programs and macros used by study team., avatar=Image{width=150,height=150,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/PfMxMarkentive/authors/kurt-preugschat.webp',altText='',fileId=null}, linkedin=https://www.linkedin.com/in/kurt-preugschat-97298923a, hs_name=, hs_path=, lastname=Preugschat, hs_initial_published_at=1716136551646, hs_created_at=1709645745122, hs_is_edited=false, hs_deleted_at=0, name=Kurt, job=, slug=kurt-preugschat, email=, hs_updated_by_user_id=9297597}, second={}, third={}})

Kurt Preugschat
has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778891, hs_child_table_id=0, hs_updated_at=1709645745122, hs_published_at=1752765086448, description=Kurt Preugschat has over 11 years of statistical experience from Phase I to Phase IV clinical trials. He is a lead statistician, providing statistical support and oversight for numerous clinical trials associated with new drug applications (NDA), supplemental new drug applications (sNDA), and medical devices. His expertise includes the development of study designs to meet complex clinical questions and the measurement of desired outcomes. In addition, Kurt has extensive experience generating statistical analyses and creating complex SAS programs and macros used by study team., avatar=Image{width=150,height=150,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/PfMxMarkentive/authors/kurt-preugschat.webp',altText='',fileId=null}, linkedin=https://www.linkedin.com/in/kurt-preugschat-97298923a, hs_name=, hs_path=, lastname=Preugschat, hs_initial_published_at=1716136551646, hs_created_at=1709645745122, hs_is_edited=false, hs_deleted_at=0, name=Kurt, job=, slug=kurt-preugschat, email=, hs_updated_by_user_id=9297597}, second={}, third={}})

Kurt Preugschat

has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778891, hs_child_table_id=0, hs_updated_at=1709645745122, hs_published_at=1752765086448, description=Kurt Preugschat has over 11 years of statistical experience from Phase I to Phase IV clinical trials. He is a lead statistician, providing statistical support and oversight for numerous clinical trials associated with new drug applications (NDA), supplemental new drug applications (sNDA), and medical devices. His expertise includes the development of study designs to meet complex clinical questions and the measurement of desired outcomes. In addition, Kurt has extensive experience generating statistical analyses and creating complex SAS programs and macros used by study team., avatar=Image{width=150,height=150,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/PfM%20Website/PfMxMarkentive/authors/kurt-preugschat.webp',altText='',fileId=null}, linkedin=https://www.linkedin.com/in/kurt-preugschat-97298923a, hs_name=, hs_path=, lastname=Preugschat, hs_initial_published_at=1716136551646, hs_created_at=1709645745122, hs_is_edited=false, hs_deleted_at=0, name=Kurt, job=, slug=kurt-preugschat, email=, hs_updated_by_user_id=9297597}, second={}, third={}})

Kurt Preugschat
has not third author: true, (SizeLimitingPyMap: {main={hs_id=187570680222, hs_child_table_id=0, hs_updated_at=1742413198455, hs_published_at=1752765086448, description=Christine has been ensuring high performance delivery of clinical research projects for more than 23 years. During this time, she has successfully led diverse teams across various cancer indications including renal, pancreatic, melanoma, lung, head and neck, breast, multiple myeloma, extra-nodal NK/T lymphoma, non-Hodgkin’s lymphoma, and Hodgkin’s lymphoma. Within the CRO setting she also supported the strategic development of over 50 hematology/oncology programs including complex Phase 1 projects in immuno-oncology and autologous cell therapy. She also led the establishment of a novel oncology research infrastructure which accelerated study activations for Phase 1 solid tumor studies. Christine obtained her master’s degree in public health at the University of Texas where she also trained in the development and implementation of clinical and epidemiologic research at the Coordinating Center for Clinical Trials as well as the Center for Occupational and Environment Health., avatar=Image{width=1200,height=1200,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Christine%20Lusk-1.png',altText='Christine Lusk-1',fileId=187579105870}, lastname=Lusk, hs_initial_published_at=1742410730688, hs_created_by_user_id=26433386, hs_created_at=1742410650472, hs_is_edited=false, hs_deleted_at=0, name=Christine, job=Director, Project Management, slug=christine-lusk, hs_updated_by_user_id=26433386}, second={hs_id=190691157084, hs_child_table_id=0, hs_updated_at=1748379058655, hs_published_at=1752765086448, description=Over the last 15 years, Megan has played a key role in advancing multiple oncology compounds through various stages of development, across numerous tumor types. Having worked at both biotech and CRO companies, Megan brings a comprehensive understanding of clinical operations and drug development. Megan maintains a focus on accelerating the development of innovative cancer therapies to improve patient outcomes worldwide., avatar=Image{width=3024,height=4032,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Megan%20Knoch.jpg',altText='Megan Knoch',fileId=190688034156}, lastname=Knoch, hs_initial_published_at=1748379090824, hs_created_by_user_id=26433386, hs_created_at=1748379018316, hs_is_edited=false, hs_deleted_at=0, name=Megan, slug=megan-knoch, hs_updated_by_user_id=26433386}, third={}})

Christine L.

Megan K.

has not third author: true, (SizeLimitingPyMap: {main={hs_id=159488778858, hs_child_table_id=0, hs_updated_at=1744830865620, hs_published_at=1752765086448, description=Rob Maiale is a marketing strategist and creative technologist with 17 years of experience turning complex ideas into growth. He currently leads content strategy, where he transforms insights from Precision experts into market-shaping narratives that drive the advancement of next-generation therapies. Rob’s career spans journalism, advertising, and brand storytelling, with a through-line of making the meaningful memorable. His work blends creative strategy with emerging AI tools to help Precision stay ahead of the curve—and above the noise., avatar=Image{width=400,height=400,url='https://5014803.fs1.hubspotusercontent-na1.net/hubfs/5014803/Rob%20Maiale.jpg',altText='Rob Maiale',fileId=184765575245}, linkedin=https://www.linkedin.com/in/rob-maiale/, hs_name=, hs_path=, lastname=Maiale, hs_initial_published_at=1744843965759, hs_created_at=1709645745089, hs_is_edited=false, hs_deleted_at=0, name=Rob, position=Associate Director, Digital Content & Inbound Marketing, job=, slug=rob-maiale, email=, hs_updated_by_user_id=9297597}, second={}, third={}})

Rob Maiale